Develop and make broadly available a robust, economically viable, shared-access platform for the technical development, manufacturing, and characterization of AAV-based gene therapy vectors. Ensure that workforce training for this platform can integrate into existing curriculum or training models to encourage adoption, and integrate improvements into the platform based on feedback from these training programs.
A gene-based therapeutics industry capable of serving patients across the full spectrum of unmet needs—from prevalent indications to ultra-rare diseases—that has access to high-quality viral vectors without cost or speed limitations.
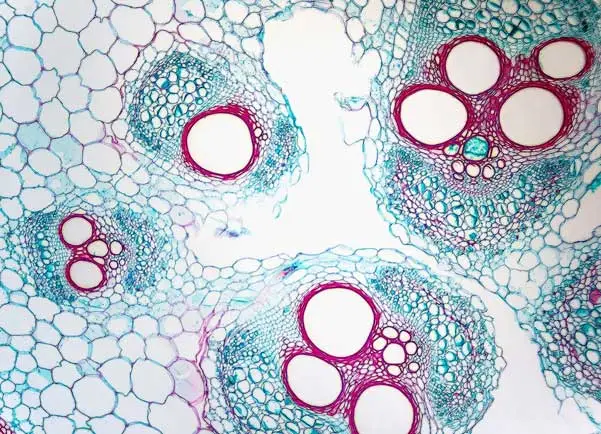
One of the most important vector platforms in use in the biopharmaceutical industry today is adeno-associated virus (AAV) vectors. Our Program focuses on advancing the manufacturing process and analytical capabilities for AAV vectors. The advances are critical for broad and rapid expansion of the emerging industry of gene-based therapeutics and ultimately the many patients and families who can benefit from access to these novel therapies.
NIIMBL is well-positioned to bring together the right resources to accelerate development of core manufacturing process platforms and essential analytical capabilities for viral vector product and process characterization, which can
The work of the program is organized around workstreams, topical focus areas.
The Translational Network Team (TNT), a core workstream within NIIMBL’s Viral Vector program, was established to align scientific innovation with the practical needs of rare and ultra-rare disease communities. TNT begins by identifying areas of highest unmet need and analyzing market trends to guide strategic focus and anticipate shifts in the gene therapy landscape. By characterizing key stakeholders, including patients, caregivers, clinicians, and advocacy groups, the team ensures that development efforts are informed by a clear understanding of who will benefit most. A major initiative within TNT is the creation of a distributed Viral Vector testbed, enabling researchers to bring rare disease vectors into larger-scale production environments. This infrastructure supports broader access to manufacturing capabilities and accelerates the translation of promising therapies.


Publication in National Academy of Engineering (NAE) The Bridge.
An Open-Access Platform: A NIIMBL Approach to Gene Therapy for Rare Diseases

Joint leadership teams Program Planning meeting

Formation of Steering Committee and Analytics and Process Leadership Teams
June 2025: NAE The Bridge: An Open-Access Platform: A NIIMBL Approach to Gene Therapy for Rare Diseases
November 2025: Voice of the Customer Survey

ABL
Alexion Pharmaceuticals

Alliance for Regenerative Medicine

AstraZeneca

Boehringer Ingelheim

Cytiva

EMD Millipore Corporation

Federal Stakeholder: National Institute of Standards and Technology

Federal Stakeholder: Food and Drug Administration

Forge Biologics, Inc.

Genentech, Inc.

Joan & Sanford I. Weill Medical College of Cornell University

Landmark Bio

Lonza Biologics, Inc.

NIIMBL

North Carolina State University

Roche

Sartorius Stedim

United States Pharmacopeial Convention

University of Florida

University of Massachusetts System

Virica Biotech Inc.

WhiteLab Genomics
To access more program information, including:
Learn more about how being a member of NIIMBL can grant you and your company access to NIIMBL-led programs and other collaboration opportunities.